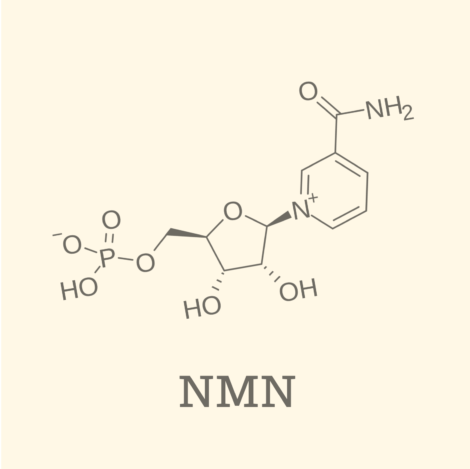
Recent years,NMN (beta-nicotinamide mononucleotide)The regulatory environment surrounding the "Mere Old Man" is changing on a global scale. In the U.S. and Japan in particular, important developments have been identified that directly impact the dietary supplement market and OEM manufacturing and ingredient supply strategies.
This article will provide an overview of the latest trends in both countries that should be kept in mind as of 2026, and we hope that understanding the regulations will be helpful in product design for companies considering manufacturing or selling NMN supplements overseas. We will now explain the trends for Q1 2026.
アメリカ:従来の医薬品扱いから、サプリメントとしての扱いに
FDA's official position
On September 29, 2025, the U.S. Food and Drug Administration (FDA) issued an official statement regarding the treatment of NMN. Its contents are,NMN is not automatically excluded from the definition of Dietary Supplement."It is called.
Around 2022, there was a widespread view in the industry that NMN could not be sold as a dietary supplement because it was the subject of pharmaceutical research. This announcement has been noted as a major shift from that trend.
The FDA cited the fact that NMN was already distributed as a dietary supplement in the U.S. and the reevaluation of the applicable conditions of the Drug Exclusion Clause (DEC) as background for this decision. This has created an environment conducive to the return of dietary supplements, including NMN, to the market.
Compliance with NDI notification system
For the sale of NMN supplements in the United States,New Dietary Ingredient (NDI) notification systemAs NMN is a relatively new ingredient, manufacturers and ingredient suppliers are increasingly submitting NDI Notifications (NDINs) and notifying the FDA of safety data and manufacturing process information.
Evidence to be maintained
In the U.S. market, not only the availability of sales,What evidence is being maintained?will be emphasized. The main points that suppliers and sales channels will ask you to confirm are as follows
- Control of origin, purity and impurities of raw materials
- Stability Tests Conducted
- GMP-compliant manufacturing system
- Labeling consistency
Even when looking to sell through e-commerce channels such as Amazon or through retail distribution, the availability of quality certification and compliance support tends to influence adoption decisions.
Points of differentiation
Based on the above, the differentiating factors in the U.S. market are as follows
- Status of NDI notification submissions
- Raw material production method (fermentation-derived, chemically synthesized, etc.)
- Purity and quality control (COA/GMP)
- Conformity of labeling and advertising wording
This regulatory arrangement will allow NMN supplements to beRe-launch and expand e-commerce salesFor companies considering OEM manufacturing or raw material supply, product design, maintenance of quality materials, and management of appealing expressions based on NDI compliance will be important issues.
Japan: Progress in Safety Evaluation of Fermentation-derived NMN
On February 4, 2026, the Food Safety Commission willβ-NMN produced using a specific microbial strain (STC2208)The results of the safety evaluation were released for the
In this evaluation, even if the production process uses genetically modified microorganisms, if the end product is highly purified,Safety is confirmed to be equivalent to that of conventional food ingredientsThe decision was made.
The use of NMN raw materials is also increasing in terms of the number of filings as "foods with functional claims," and opportunities in the domestic market are expanding.
Key Points in OEM Development
In light of the respective regulatory trends in the U.S. and Japan, the following points require special attention in the development of NMN products.
- for the U.S.: Product design based on NDI notification and labeling compliance
- for JapanSafety confirmation of raw materials and conformity to labeling
- be commonTransparency of raw material production process, including NMN derived from fermentation
- Export supportQuality control system including: GMP compliance, Certificate of Analysis (COA), etc.
The NMN market is expected to continue to grow, and raw material quality and regulatory compliance are factors that determine the competitiveness of OEM companies. When considering overseas sales, companies must have not only manufacturing capacity, but also a system that can consistently handle raw material selection, document preparation, and labeling design based on an understanding of regulations.
For those considering OEM manufacturing of NMN supplements
ZaaZ Inc. provides consultation regarding OEM manufacturing of NMN supplements. We will propose product development according to your objectives, including formulation design, raw material selection, production lot, and package specifications. Please feel free to contact us for a consultation.
Contact us